Selling kombucha across borders isn’t just about brewing a great SCOBY, it’s about proving your culture won’t land you in regulatory hot water. A Canadian kombucha brewery learned this the hard way in 2024 when a shipment bound for California was held at the border. The issue? Their product tested at 0.6% ABV—legal in Alberta at 1.1%, but strictly over the 0.5% U.S. federal limit.
If you’re operating a commercial kombucha facility anywhere in the world, you need to answer one question before filling your first bottle: Which market are you selling into? The answer determines everything from your pH monitoring frequency to whether you need a HACCP plan, an ISO 22000 certificate, or a full liquor license.
The Regulatory Reality: One Product, Multiple Rulebooks
Kombucha occupies a unique position in global food law—classified as a non-alcoholic fermented beverage in most jurisdictions, but subject to wildly different safety thresholds. Unlike carbonated soft drinks with fixed formulas, kombucha continues fermenting after packaging. This biological reality creates compliance challenges that static regulations struggle to address.
Here’s what you’re dealing with: alcohol thresholds range from 0.05% ABV in the UK to 1.2% in parts of the EU, with pH requirements, labeling mandates, and certification needs varying just as dramatically. A product legally sold as “non-alcoholic” in Sydney could require alcohol excise tax labeling in London and face outright rejection in Tokyo.
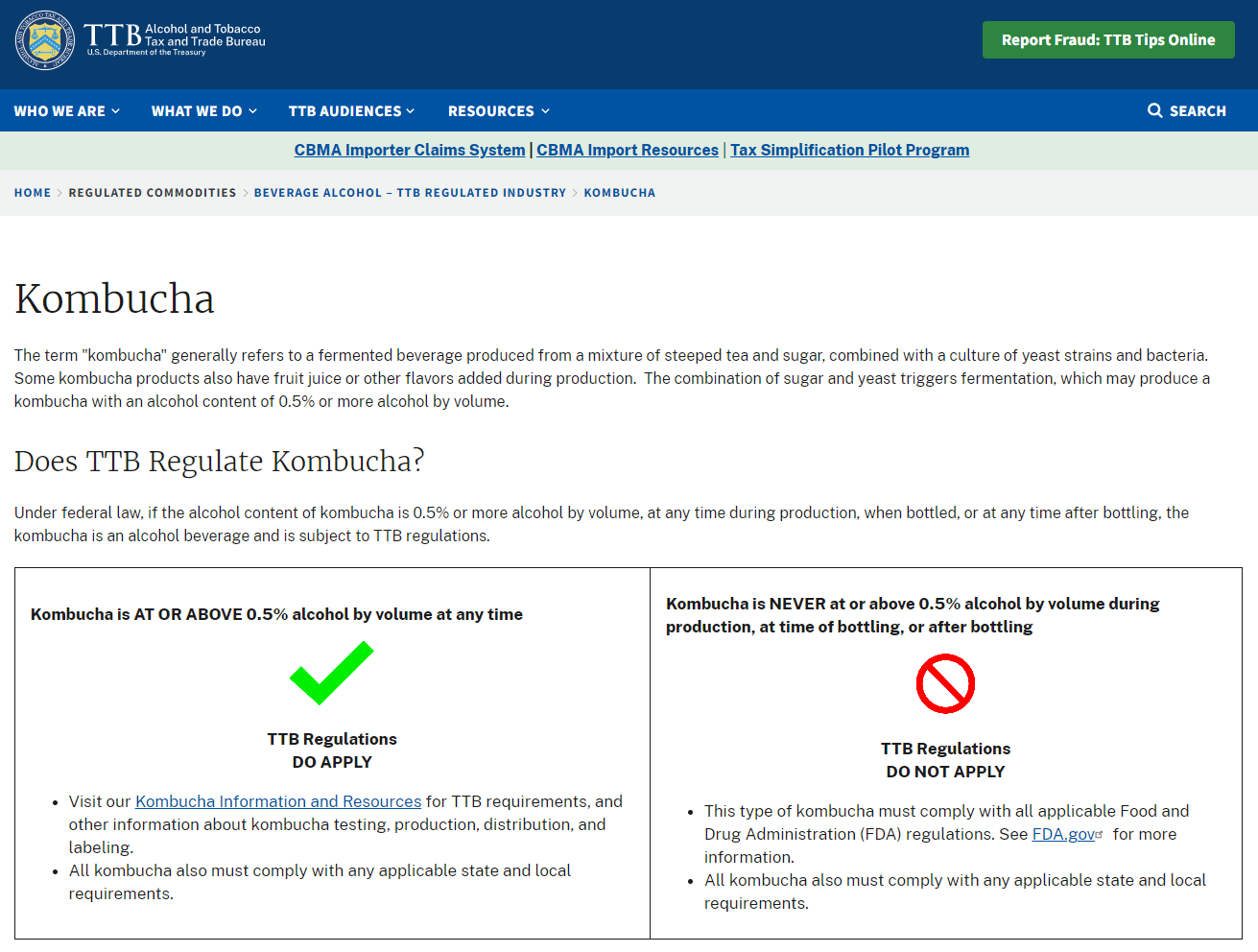
United States: The Strictest Common Denominator
The U.S. Food and Drug Administration (FDA) set the global benchmark for kombucha regulation, primarily because it’s the world’s largest single market and the most restrictive major jurisdiction.
The 0.5% ABV Red Line: Under the Federal Alcohol Administration Act, beverages containing ≥0.5% alcohol by volume are subject to the Alcohol and Tobacco Tax and Trade Bureau (TTB), which requires formula approval, health warnings, age-restricted distribution, and excise taxes. Below this threshold, FDA regulates kombucha as a food product subject to the Food Safety Modernization Act (FSMA) preventive controls.
Critical Control Requirements:
- pH ≤2 at packaging to prevent pathogen growth
- HACCP-based food safety plan mandatory under FSMA preventive controls
- Foreign Supplier Verification Program (FSVP) compliance if you’re exporting to the U.S.—your American importer must verify your facility meets U.S. safety standards
- Process Authority Letter required if adding low-acid ingredients post-fermentation
For international kombucha breweries, the message is clear: target 0.5% ABV maximum if you want seamless U.S. market access. Some producers aim for ≤0.3% to build in safety margins against post-bottling fermentation creep.
European Union: Fragmented Federalism
The EU presents a patchwork where Brussels sets baseline food safety law, but member states enforce wildly different alcohol thresholds.
| Jurisdiction | Non-Alcoholic Threshold | Notes |
| EU General (EFSA) | ≤0.5% ABV standard | Novel Foods Regulation compliance; mandatory ingredient/nutrition labeling |
| Denmark, Lithuania, Luxembourg, Slovenia, Spain | ≤1.2% ABV | “Low-alcohol” classification; alcohol % must appear in nutrition panel |
| Finland | ≤1.2% ABV (but 2.8% for marketing) | 1.2-2.8% pays alcohol taxes but can be marketed as “non-alcoholic”—no alcohol beverage marketing allowed unless below 2.8% |
| Slovakia | ≤0.75% ABV | Strictest in Central Europe |
| United Kingdom | ≤0.05% ABV for “alcohol-free”; ≤1.2% for “low-alcohol” | Post-Brexit divergence from EU; “non-alcoholic” cannot be used with alcohol-associated names |
The HACCP Mandate: Unlike the U.S., where HACCP is effectively required through FSMA, the EU explicitly mandates HACCP implementation under Regulation (EC) No 852/2004. This isn’t optional—it’s inspected and enforced. Many European retailers, particularly in the UK, now require BRCGS (British Retail Consortium Global Standards) certification as a condition of supply.
Strategic Implication: If you’re targeting EU-wide distribution, brewing to ≤0.5% ABV eliminates regulatory friction. The 1.2% “low-alcohol” category exists but creates labeling complexity and potential consumer confusion.
Canada: Provincial Patchwork North of the Border
Canada’s federal system creates compliance complexity that rivals the EU. Health Canada sets baseline standards, but provinces enforce specific alcohol thresholds.
| Province | Non-Alcoholic Threshold | Notes |
| Alberta, British Columbia, and Manitoba | ≤1.1% ABV | “Low alcohol” acceptable claim |
| Nova Scotia, Ontario, PEI, Quebec, Saskatchewan | ≤0.5% ABV | Stricter alignment with the U.S. standard |
Labeling Requirements: Unlike the U.S., Canada mandates alcohol content declaration on labels if the product exceeds 0.5% ABV—even if it’s below the provincial non-alcoholic threshold. This creates transparency but requires careful label management for multi-province distribution.
Health Guidance: Health Canada advises pregnant women and adolescents to avoid all kombucha regardless of alcohol content—a conservative stance that affects market positioning.
Certification Path: While HACCP isn’t federally mandated, the British Columbia Centre for Disease Control (BCCDC) explicitly recommends HACCP implementation for commercial kombucha production, emphasizing pH and alcohol monitoring as critical parameters. Major Canadian retailers typically require HACCP or ISO 22000 certification.
Australia & New Zealand: The 1.15% Outlier
Australia and New Zealand operate under the Food Standards Australia New Zealand (FSANZ) Code, which takes a distinctly different approach to fermented beverages.
The 1.15% ABV Standard: Under Standard 2.6.2, a “brewed soft drink” (including kombucha) can contain up to 1.15% ABV and still be classified as non-alcoholic. This is the highest major-market threshold globally and reflects a regulatory philosophy that acknowledges natural fermentation byproducts.
The State-Level Catch: Australian states enforce divergent standards :
- Queensland, Tasmania, Victoria: >0.5% ABV requires alcohol labeling and is treated as liquor under state law
- Western Australia, South Australia, New South Wales: The full 1.15% FSANZ standard applies
Labeling Mandate: Any beverage containing >0.5% ABV must declare alcohol content and standard drink information. A 2023 national survey found 65% of tested kombucha samples exceeded 0.5% ABV, with many non-compliant on labeling.
Emerging Licensing Requirements: A significant 2024 development—Australia is proposing a Fermented Beverage License that would require HACCP or FSSC 22000 certification for all producers. This would align Australia with EU-style mandatory food safety management systems.
Asia-Pacific: Emerging Markets, Evolving Standards
- Japan: The Ministry of Health, Labor and Welfare (MHLW) classifies kombucha as a “health food” under the Food Sanitation Act. While HACCP isn’t legally mandated, the probiotic health claims that drive Japanese kombucha sales require rigorous substantiation. Alcohol limits align with the 0.5% general non-alcoholic standard, but enforcement focuses on microbiological safety and labeling accuracy.
- South Korea: The Ministry of Food and Drug Safety (MFDS) regulates kombucha as a functional health food. This classification requires stricter facility inspections and documentation than general food production. Sales of fermented health beverages grew 10% annually between 2020-2024, drawing increased regulatory scrutiny.
- India: The Food Safety and Standards Authority of India (FSSAI) mandates probiotic food standards requiring label declaration of viable counts and strain identification. Kombucha falls under the fermented/functional beverage classification, with FSSAI licensing required. The tropical climate creates unique challenges—temperature variability during fermentation can lead to alcohol spikes and inconsistent quality, making HACCP implementation critical for export credibility.
- China: National beverage standards classify products with ≤0.5% alcohol mass fraction as non-alcoholic. However, enforcement varies by region, and imported kombucha faces strict customs inspection for microbiological contamination and alcohol content verification.
- UAE/GCC: The UAE Food Safety Regulations (2020) establish comprehensive kombucha standards, mandating GMP compliance, cold chain maintenance, and regular microbiological testing. The 0.5% ABV threshold applies, with strict labeling requirements for ingredients and fermentation processes.
The Certification Hierarchy: From HACCP to FSSC 22000
Regardless of your market, you’ll face questions about food safety certification. Here’s how the options stack up for kombucha producers:
The 2024 Shift: A notable trend—FSSC 22000 is rapidly becoming the gold standard for international kombucha trade. Finnish producer The Good Guys explicitly pursued FSSC 22000 certification in 2025 to “strengthen quality, improve food safety management throughout the production chain, and enable growth in export markets”. This reflects market reality: major retailers and distributors increasingly require GFSI-recognized certification as a risk mitigation tool.
For equipment manufacturers and co-packers, FSSC 22000 or ISO 22000 certification is becoming a prerequisite for partnership. As one Vietnamese supplier noted, “Certifications such as HACCP, BRCGS, and GMP are increasingly required by retailers, prompting producers to invest in compliant facilities—a factor reflected in pricing”.
Strategic Recommendations by Business Model
If You’re a Startup Kombucha Brewery (Local/National Focus):
- Implement HACCP immediately—it’s the global baseline and often legally required
- Target ≤0.5% ABV regardless of local limits to preserve future export optionality
- Invest in validated pH monitoring (digital meters, calibration logs) from day one
If You’re Expanding Internationally:
- Conduct market-specific regulatory mapping before production begins
- Consider ISO 22000 certification to satisfy multiple import jurisdictions efficiently
- Establish separate production protocols for strict markets (U.S., UK) versus tolerant markets (Australia, parts of EU)
If You’re a Contract Manufacturer/Co-Packer:
- Pursue FSSC 22000 to attract premium international clients
- Maintain alcohol testing capabilities in-house; don’t rely on third-party labs for routine batch verification
- Document everything—importers will need records for their FSVP compliance
If You’re Exporting to the U.S. as a Foreign Facility:
- Verify your U.S. importer has a robust FSVP program—they’re legally liable for your compliance
- Maintain English-language batch records, corrective action logs, and supplier documentation
- Expect FDA inspections; the agency has increased foreign facility surveillance since 2023
From Compliance Knowledge to Production Capability
Understanding global regulations is the first step. Building production systems that satisfy them consistently is the second—and it’s where most ambitious kombucha breweries stumble.
You can map every alcohol threshold and pH requirement on paper, but if your fermentation vessels can’t hold temperature within ±0.5°C across a 10,000L batch, you’ll produce variance that no amount of documentation can explain away. If your CIP system can’t achieve a 3-log reduction validated for your specific SCOBY strain, your HACCP plan remains theoretical.
At Tiantai, we engineer around compliance reality, not catalog standards. No two kombucha operations face identical regulatory matrices—your equipment shouldn’t pretend otherwise. We design tank configurations, jacketing systems, and CIP protocols specifically for your target markets: whether that’s U.S. FDA preventive controls, EU HACCP with BRCGS retail requirements, or Australia’s emerging fermented beverage licensing standards.
Your personalized equipment drawings are ready within 7 days. Your production reality follows within 60 days, with the 20-year tank integrity that outlasts regulatory changes in any jurisdiction you enter.
Because global market access isn’t just about knowing the rules. It’s about equipment that lets you play with them on a scale.